The technology of supercapacitors and batteries varies widely. There are advantages and disadvantages to both technologies, despite how the part is constructed or the materials used in creating the part. From an application standpoint, there may be some applications that currently use batteries where supercapacitors can replace them, but most of those applications require batteries or a hybrid configuration, which utilizes both technologies.
Supercapacitors are similar to other types of capacitors because they serve the same function—charging and discharging energy. The difference is how much energy they can store and at what voltages they operate. These variations are caused by the differences in how supercapacitors are designed compared to other capacitor technologies. The dielectric in ceramic, tantalum, and aluminum capacitors contains hundreds to thousands of molecules whereas supercapacitors use one molecule to form the dielectric. In KEMET’s supercapacitors, this molecule is a highly activated carbon, which is coated on the aluminum electrode. The dielectric is formed when the electrolyte comes into contact with the carbon and forms what is called the inner Helmholtz Layer. The outer Helmholtz layer shown in Figure 1 serves as the cathode of one of the capacitors on one electrode and one of the capacitor’s anodes on the other electrode. Capacitors (in plural form) is mentioned because two capacitors are formed with this structure.
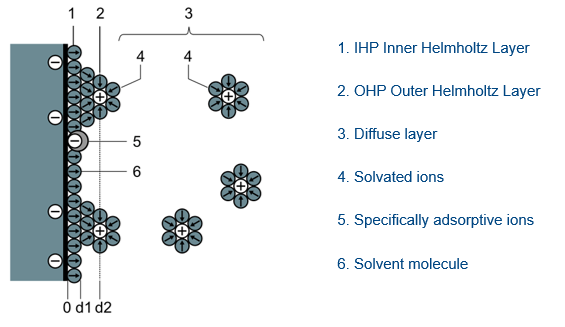
In this formation, the outer Helmholtz layer, which creates both cathode and anode can be confusing. The outer Helmholtz layer forms one cathode and one anode, while the other cathode and anode are formed on both sides of the separator when the capacitor is charged up. This is where the name EDLCs, electric double layer capacitors, comes from. Both electrodes are covered with the thin layer of carbon along with the electrolyte, which has a separator in between. This forms the double layer and can be seen in Figure 2 that there are two capacitors.
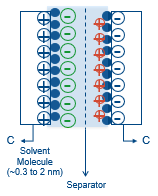
These structural differences lead to two results: higher capacitances and lower operating voltages, when compared to other types of capacitors. The higher capacitance is achieved by having a carbon that is a very porous material. This increases the surface area, which is proportional to the capacitance. Since there is only one molecule, the electrolyte is very thin, which means the distance between the electrodes is much smaller than other technologies. This allows the part to have a higher capacitance as the distance between electrodes is inversely proportional to the capacitance as shown in the equation below
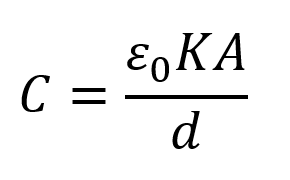
This also explains why the voltage rating of supercapacitors is low. Since the dielectric is one molecule thick, voltages higher than the rated voltage place electrical stress across the electrolyte molecule and tears it apart. This damage is irreversible and the part cannot self-heal like other capacitor dielectrics.
There are four main differences between supercapacitors and batteries: energy density, power density, lifetime, and cost.
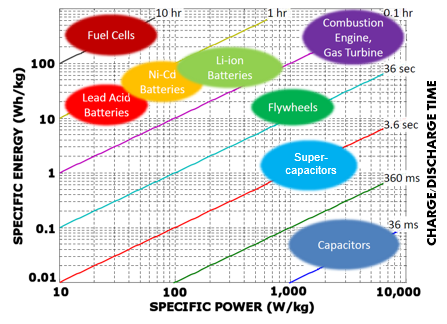
Energy density refers to the amount of charge a technology can hold. As shown in Figure 3, capacitors have the lowest energy density of commonly used storage devices. Supercapacitors have the greatest energy density of any capacitor technology, but batteries are far superior than any capacitor in this category. Batteries store charge chemically, while capacitors store charge electrically. Chemical reactions have the capability to store much more energy than electrical storage, which is what contributes to batteries being used more often in applications that require higher storage.
Power density refers to the amount of time it takes a technology to charge and discharge energy. Batteries have the disadvantage in this characteristic due to the chemical reactions that take place to store and release energy. Supercapacitors have faster charge and discharge rates than batteries because the chemical reactions that take place within batteries take longer to release electrons than the electrical discharge in supercapacitors.
Chemical reactions are the limiting factor for the lifetime of batteries. In batteries, there are two different materials used as the electrodes, one for the anode and the other for the cathode. The electrolyte reacts with both electrodes and provides the chemical reactions, which produce protons (positive ions) at the positive electrode and electrons (negative ions) at the negative electrode. This causes different chemical reactions to occur at the electrons, one that gives up ions and the other that accepts ions. Once this electrolyte is
used up, no chemical reactions can occur, and the battery stops working because it cannot store or discharge any longer. The number of cycles is much smaller than that of supercapacitors because capacitors do not rely on chemical reactions to store energy making the lifetime of supercapacitors much longer than batteries.
Supercapacitors have a much higher up-front cost than batteries, which causes many designs to use batteries instead. Given the differences in lifetime of supercapacitors and batteries, the long-term cost of supercapacitors may be a cheaper option even with the higher initial cost. It all depends on the lifetime needed for the specific application.
In many applications, the differences mentioned previously rule out replacing batteries with supercapacitors. Some applications do not have the space to add as many supercapacitors as it takes to reach the energy density that batteries contain. Other applications need the supercapacitors to hold the charge longer or they have to be recharged more often than batteries, which is not always an option. There are advancements in supercapacitor technologies that allow a higher energy density, but this capability is still not within the range of a battery’s energy density. In some applications though, a hybrid configuration prove to be the most useful. The supercapacitors provide the quick burst of energy for an application, while the batteries handle the long-term energy needs.